Insider Brief
- Researchers synthesized and imaged a carbon-based molecule whose electron system adopts a previously unrealized half-Möbius topology, meaning the electron orbitals twist by about 90 degrees as they travel around the ring.
- Using scanning probe microscopy and controlled voltage pulses, the team directly observed the molecule’s twisted electronic structure and reversibly switched it between two mirror-image half-Möbius states and a conventional planar state.
- The results show that quantum topology can be engineered and controlled at the level of a single molecule, opening new avenues for studying how twisted electronic structures affect magnetic, electronic and quantum properties.
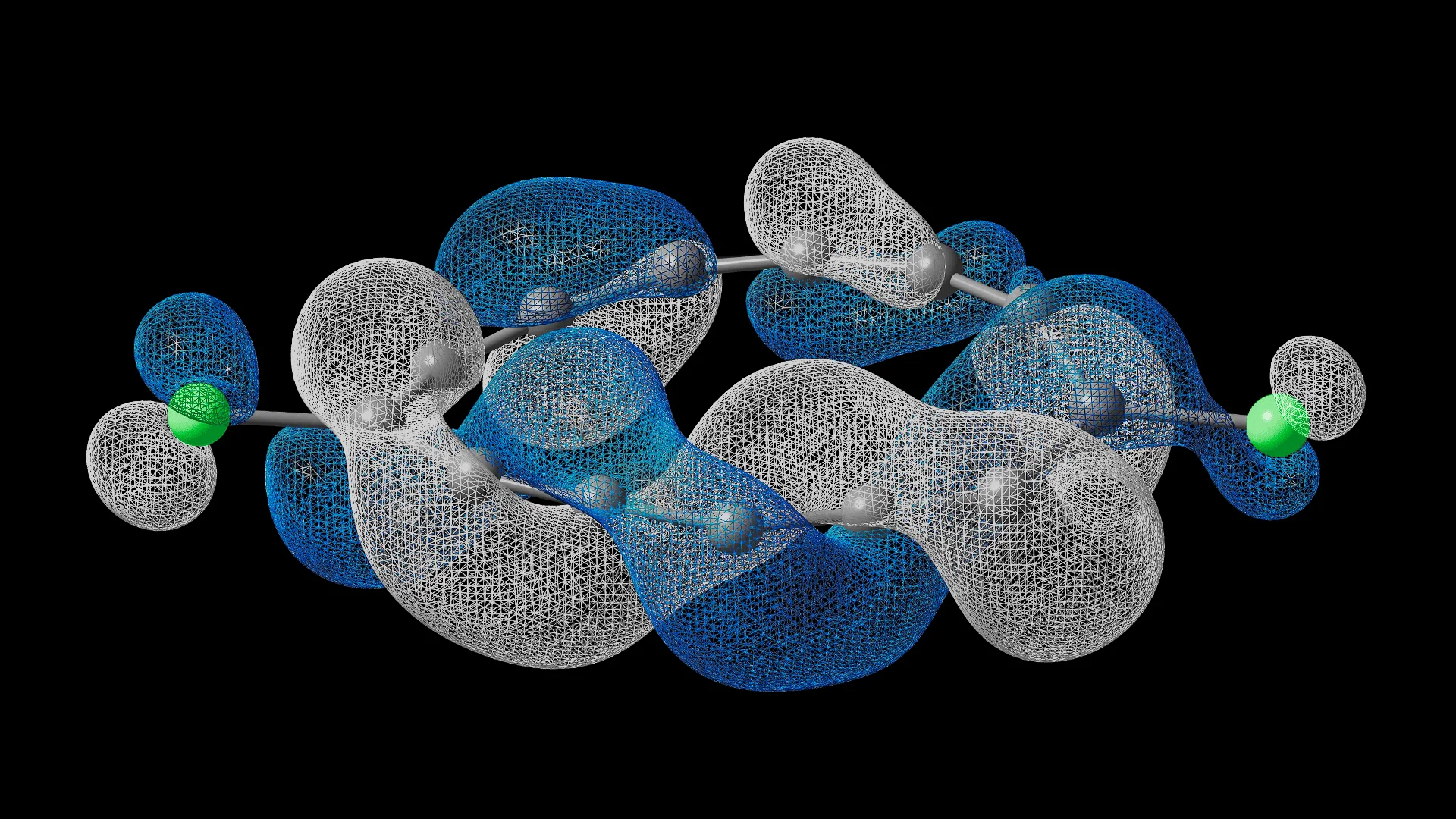
- Dyson orbital for electron attachment, calculated using quantum hardware. (Image: IBM Research and the University of Manchester)
A team of chemists built and directly imaged a molecule with electrons that move as if they are traveling along a half-twisted Möbius strip — and then switched it back and forth between distinct quantum states.
In a study published in Science, a researcher team, including IBM Quantum scientists, report on the carbon ring with electron cloud that forms — what they describe as — a “half-Möbius” topology. The work extends decades of theory about twisted molecular systems and demonstrates that electronic topology — the way quantum wavefunctions wrap and connect — can be engineered, observed and controlled at the level of a single molecule.
The finding adds a new entry to the short list of molecules whose electronic structure is defined not just by which atoms are bonded, but by how quantum phases accumulate as electrons circulate around a ring. Importantly, for the quantum community, an IBM quantum computer helped confirm their calculations.

“First, we designed a molecule we thought could be created, then we built it, and then we validated it and its exotic properties with a quantum computer,” said Alessandro Curioni, IBM Fellow, Vice President, Europe and Africa, and Director of IBM Research Zurich, in a news release. “This is a leap towards the dream laid out by renowned physicist Richard Feynman decades ago to build a computer that can best simulate quantum physics and a demonstration where, as he said, ‘There’s plenty of room at the bottom.’ The success of this research signals a step towards this vision, opening the door for new ways to explore our world and the matter within it.”
A Half-Twist in Quantum Space
Most people encounter a Möbius strip as a mathematical curiosity. Just take a strip of paper, twist it once by 180 degrees, and tape the ends together. The result has only one continuous side. In chemistry, the concept has a parallel. In certain ring-shaped molecules, the cloud of electrons that sit above and below a carbon ring and enable aromatic stability — known as π-electrons — can twist as they go around the ring.
In familiar aromatic molecules such as benzene, that electron cloud forms a simple, untwisted loop. In so-called Möbius aromatic molecules, first proposed in the 1960s and realized experimentally in the early 2000s, the electron system twists by 180 degrees in one full circuit. That twist changes the phase of the electron wavefunction and alters the molecule’s stability and magnetic behavior.
This interests chemists, especially, because of they see these twisted electron systems as a way to explore unusual quantum behavior and potentially design molecules with new electronic and magnetic properties.
But, the new study goes one step further — or, maybe a bit more precisely, one half-step further.
The researchers synthesized a 13-carbon ring with two chlorine atoms attached. In this system, the π-electron framework twists by 90 degrees in a single circuit around the ring. The researchers call this arrangement a “half-Möbius” topology.
That difference may sound small, but in quantum mechanics it matters. In a full Möbius system, the electron wavefunction flips sign after one trip around the ring. In the half-Möbius system, the sign change happens only after two trips. The system returns fully to its starting condition only after four loops.
In simple terms, the electrons “remember” where they have been in a way that ordinary ring molecules do not.
Built Atom by Atom on a Surface
The molecule was not synthesized in a flask. Instead, the team constructed it on an insulating surface using a scanning probe microscope, a tool capable of moving and removing individual atoms.
The researchers began with a precursor molecule containing 13 carbon atoms and 10 chlorine atoms. By applying carefully controlled voltage pulses with the tip of a scanning tunneling microscope, they removed eight chlorine atoms one by one. The process left behind a C13Cl2 ring.
The molecule was assembled on a thin layer of sodium chloride placed atop gold. The salt layer electrically insulated the molecule from the metal beneath, allowing its intrinsic electronic structure to be studied without interference.
To determine the molecule’s shape, the team used atomic force microscopy with a carbon monoxide–functionalized tip, which can resolve the positions of individual atoms and even distinguish bond types. The images showed that the carbon ring was not flat. Instead, it adopted a slightly twisted, chiral geometry — meaning it exists in left-handed and right-handed forms.
Those subtle distortions were the first sign that the electron cloud itself might be twisted.
The most striking evidence came from scanning tunneling microscopy measurements of the molecule’s lowest unoccupied molecular orbital — effectively a map of where an added electron would go.
In the twisted singlet state, the orbital density displayed a helical pattern around part of the ring. Rather than separating neatly into “above” and “below” components, as in conventional aromatic systems, the electron cloud wrapped around the ring in a continuous twist.
However, when the molecule occupied a different electronic state — a planar triplet configuration — the orbital pattern reverted to a conventional, untwisted form. In that state, the carbon ring flattened and the electron cloud split cleanly into in-plane and out-of-plane components.
In other words, the topology of the electron system could be toggled between a nontrivial half-twist and a conventional configuration.
Switching Between Topologies
The researchers found they could switch the molecule reversibly among three configurations, specifically, a left-handed half-Möbius singlet, a right-handed half-Möbius singlet and a planar triplet state.
By increasing the voltage applied through the microscope tip, they triggered transitions between these states. Measurements of the tunneling current over time revealed distinct signatures corresponding to each configuration.
The switching threshold — roughly a few tenths of a volt — matched theoretical predictions of the energy difference between the states.
The two singlet configurations are mirror images of each other. Their geometry arises from a phenomenon known as a pseudo–Jahn-Teller effect. This effect describes that when certain electronic states interact, the molecule lowers its energy by distorting slightly. In this case, that distortion produces the chiral, twisted geometry associated with the half-Möbius topology.
When the molecule transitions to the triplet state, that distortion disappears and the ring relaxes toward a flat structure.
The importance of the work lies less in the specific molecule and more in what it demonstrates.
Topology — a branch of mathematics concerned with properties that remain unchanged under continuous deformation — has become central to modern physics. Topological insulators and superconductors, for example, have electronic states protected by global features of their wavefunctions rather than by local chemistry.
This study shows that topology can also be engineered in single organic molecules, not just in extended solids.
The half-Möbius topology imposes unusual boundary conditions on the electron wavefunction. The researchers report that it corresponds to a Berry phase — a kind of geometric phase acquired by a quantum system — that differs from both conventional and full Möbius systems. In principle, that could influence how electrons respond to magnetic fields or how currents circulate around the ring.
The calculations also suggest relatively strong spin–orbit coupling in the twisted configuration, which could make such systems interesting for studying spin-dependent effects.
Quantum Computing Meets Chemistry
To confirm their interpretation, the researchers performed advanced computer simulations to model the molecule’s electronic structure. They also tested part of the calculation on a quantum computer, using 72 qubits — making it one of the largest quantum chemistry simulations of its kind so far.
The quantum-assisted results agreed with classical calculations, reinforcing the conclusion that the molecule’s ground state indeed features a half-twisted electronic topology.
The team suggests that quantum hardware may become increasingly useful for modeling systems in which topology, strong electron correlation and geometry intertwine in ways that challenge traditional computational methods.
There are, of course, limits and plenty of space for future work. The half-Möbius molecule exists only under controlled laboratory conditions, which includes ultra-high vacuum, cryogenic temperatures and atom-by-atom assembly.
The topology is not permanently encoded in the molecular framework. It arises from a delicate balance of electronic interactions and substituent effects. That balance makes the system switchable. but also fragile.
Future work will likely explore whether similar topological features can be built into larger molecular networks or incorporated into materials where they might influence measurable properties such as magnetism or conductivity.